For over a century, the human experience of narcolepsy has been documented [2]. Yet, for many of us living with Type 1 Narcolepsy (NT1) and its constant, challenging realities, the world still dismisses our struggle as mere fatigue or laziness.
I’m here to tell you, as an independent advocate living with NT1 and two other sleep disorders, that this misunderstanding simply cannot continue. The science developed over the past three decades is incredibly valuable and informative, and it overwhelmingly supports what we feel every day: NT1 is a neurological system crisis, rooted in a neurological system dysfunction, not just a problem with sleep. Narcolepsy is defined as a primary central nervous system (CNS) disorder resulting from complex interactions of genetic, environmental, and immunological factors [1][2].
Given this profound, systemic nature – affecting the CNS, cardiovascular, metabolic, autonomic systems, and seemingly beyond – it seems out of line that NT1 remains primarily classified and seen simply as a ‘sleep disorder’. Many of us ask: At what point does this classification change? As a condition resulting from the destruction of hypocretin neurons [2], which I personally consider to be an ‘Organ-Specific Autoimmune Disorder’ causing Neurologic System Dysfunction, NT1 should be seen with a seriousness comparable to diseases like Multiple Sclerosis (MS) or Type 1 Diabetes (T1D). The pathology of narcolepsy, which involves a tight association with HLA Class I alleles, suggests a cell-mediated cytotoxic mechanism that implicates NT1 alongside other autoimmune disorders [1][4].
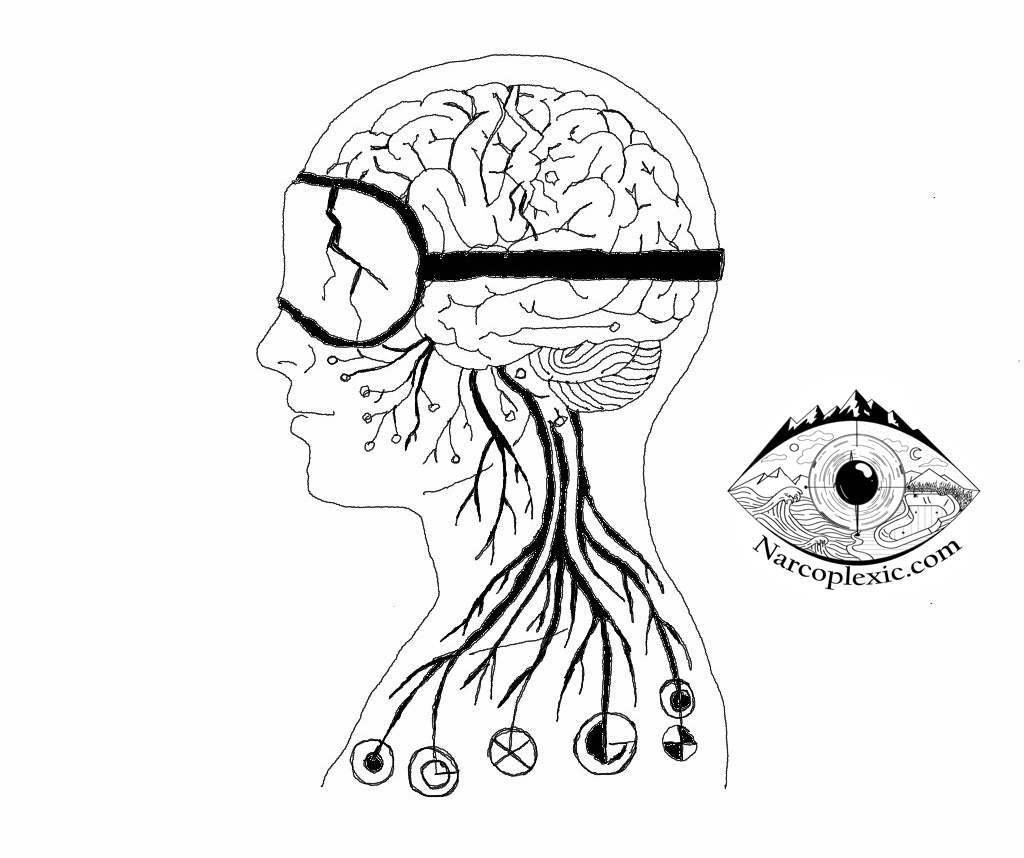
The Science Has Spoken: It’s an Autoimmune Attack
The scientific consensus has shifted dramatically since the discovery of hypocretin (also called orexin) deficiency around the year 2000 [2]. We now understand that NT1 is primarily caused by an autoimmune process – where the body mistakenly attacks and destroys the hypocretin-producing neurons deep in the hypothalamus [4][1]. This destruction leads to an irreversible loss of these cells [3].
This mechanism is why genetics are so critical. Carrying the HLA gene marker $DQB1*06:02$ makes a person highly susceptible to developing NT1 [1]. This allele is found in approximately 88–98% of patients with clear cataplexy across ethnic groups [1][5]. However, this gene is found in about 30% of the general population, while NT1 itself is rare, affecting roughly 1 in 2,500 people [2].
The genetic predisposition isn’t the whole story. Environmental factors act as the “trigger”. Medical literature confirms that infections like certain flu strains (such as H1N1), streptococcal infections, or even trauma can initiate the autoimmune reaction that wipes out those essential brain cells [2][1]. Research even suggests that the condition may involve a T-cell mediated cytotoxic mechanism (CD8+ T-cells or NK cells) [2][4], representing the final step in hypocretin neuronal destruction [2]. Given this complexity, we must ask if other profound systemic stressors, such as severe psychological or physical trauma, deep illnesses, or even behaviors like long-term inconsistent sleep patterns in youth, could also contribute to triggering the disease onset. This inquiry is especially relevant as ongoing studies explore how the gut microbiome and the microbiota-gut-brain axis play a significant role in NT1’s pathological process and causation [1][3].
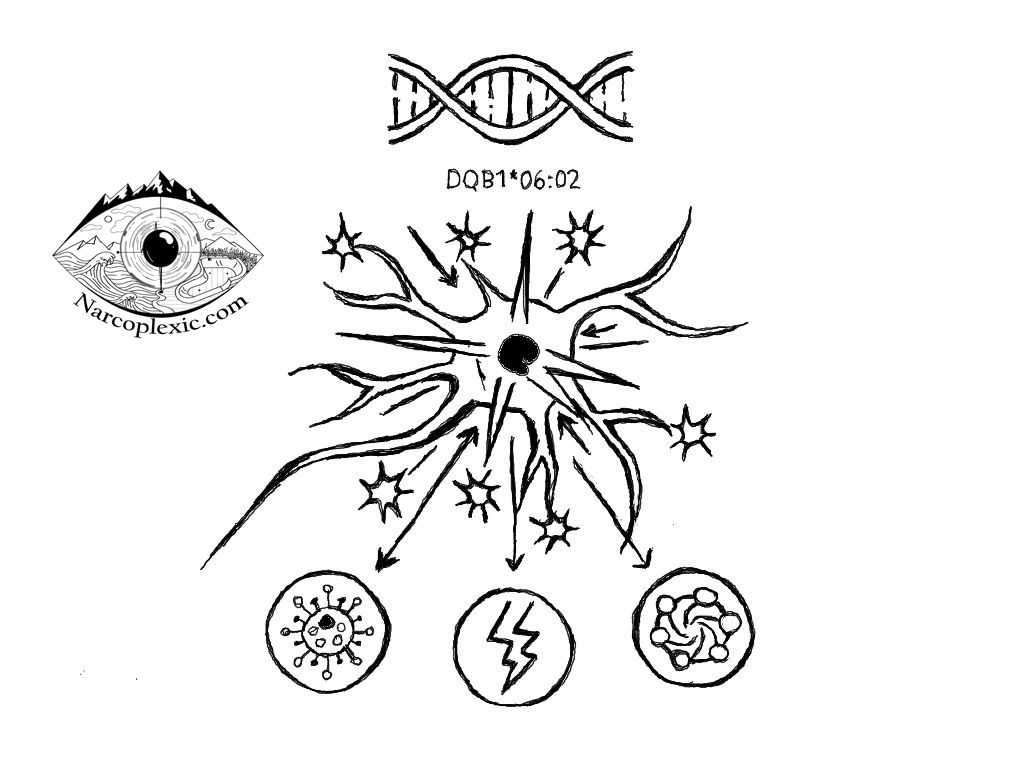
Orexin: The Conductor of Your Body’s Orchestra
The loss of hypocretin/orexin isn’t just about feeling sleepy [3]. These neuropeptides are aptly described as the “regulator/conductor of the orchestra of neurotransmitters, neuropeptides, hormones” that dictate our overall psychological and physical stability and well-being.
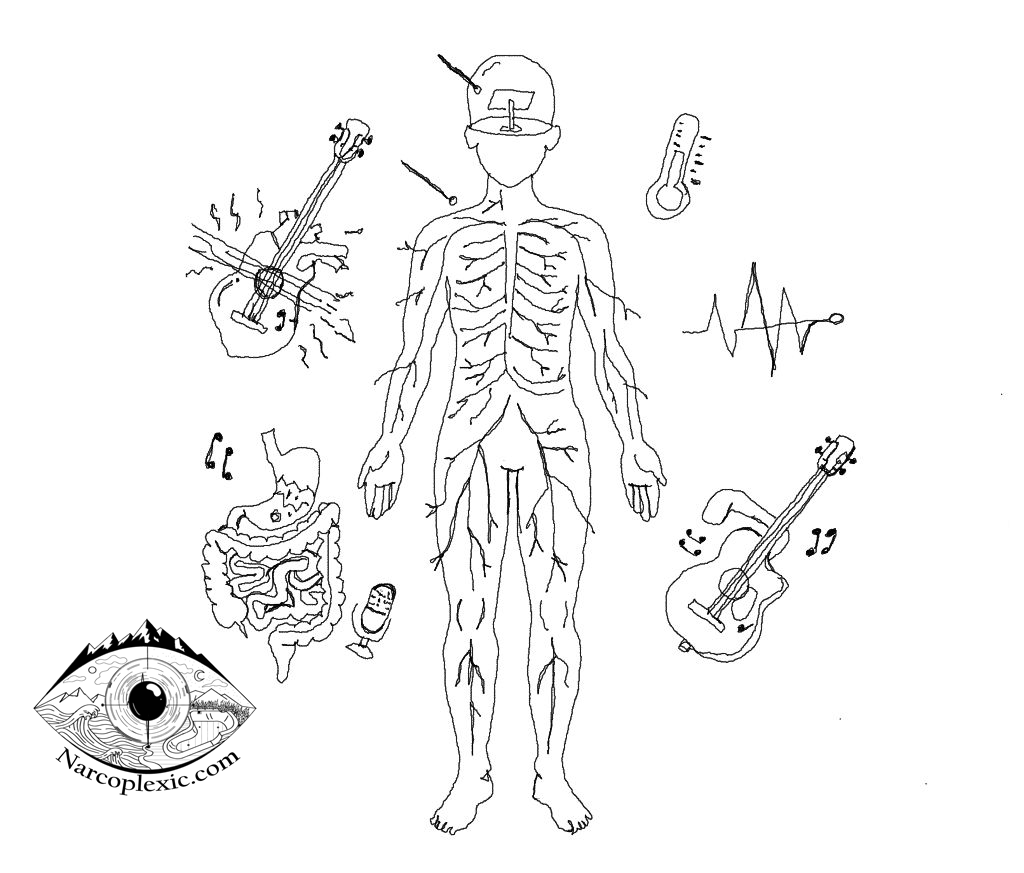
When the conductor is gone, the entire body falls into systemic dysfunction. The science clearly demonstrates that NT1 symptoms ripple far beyond excessive daytime sleepiness (EDS) into multiple bodily systems:
- The Autonomic Nervous System (Dysautonomia): Narcolepsy patients frequently suffer from clinically relevant dysautonomia [1], which is dysfunction of the body’s involuntary control systems [1]. This dysautonomia is found in nearly all domains investigated (pupillomotor, urinary, gastrointestinal, cardiovascular, and thermoregulation) except typically sexuality. This spectrum of non-motor symptoms is actually comparable to those observed in idiopathic Parkinson’s disease (IPD), suggesting wide-ranging, clinically detectable dysfunction beyond the narcoleptic core syndrome.
- Cardiovascular Challenges: Many patients experience issues like low blood pressure upon standing (orthostatic symptoms) [1] and changes in blood pressure during sleep (non-dipping BP profile) [1]. Patients with NT1 exhibit attenuated cardiovascular reflexes compared to healthy controls [1]. This systemic deregulation translates into a frightening reality: individuals with narcolepsy face a heightened risk of new cardiovascular events [1]. Issues often observed include non-dipping blood pressure profiles during sleep and reduced heart rate variability [1].
- Metabolic and Temperature Issues: The deficit disrupts energy balance, contributing to the high prevalence of obesity seen in NT1 [1]. Orexin deficiency is believed to impair Brown Adipose Tissue (BAT) functionality, worsening adiposity. Furthermore, changes in body temperature regulation occur, with daytime sleep attacks often preceded by a measurable increase in distal skin temperature and distal-to-proximal temperature gradient (DPG) [1].
- Gut-Brain Axis: Recent investigations show a clear link between NT1 and the gut microbiome. Research confirms bidirectional causal links between certain types of gut bacteria and NT1 [1][3], highlighting the importance of the microbiota-gut-brain (MGB) axis in the disease’s pathological process [1][3]. Studies have shown differences in intestinal flora structure in NT1 patients [1][3], with some evidence suggesting that an increased relative abundance of the pro-inflammatory bacterium Klebsiella [1][6] and a decreased abundance of anti-inflammatory genera like Blautia (which is inversely correlated with NT1 risk) may be involved [1][3].
The literature is clear: NT1 is a disorder that impacts our entire neurological system [1], leading to fragmented sleep [1], frequent awakenings [1], and a significantly increased struggle with depression and fatigue, regardless of sleepiness or obesity status [1].
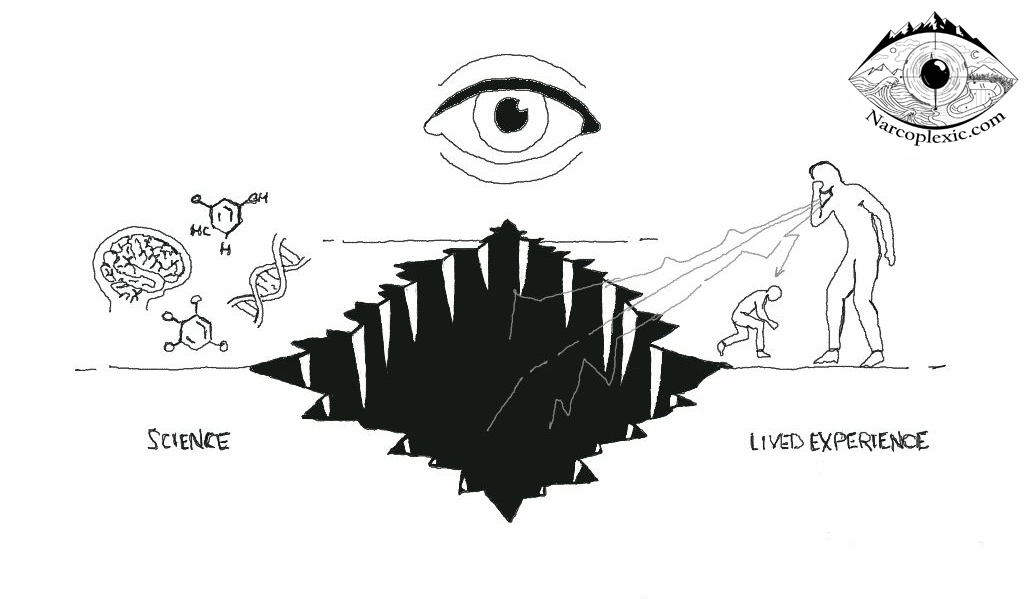
The Gap: From Scientific Fact to Human Acknowledgment
The medical literature figured out the root cause of this debilitating condition over the past 30 years. This scientific progress is hugely valuable, giving us critical insights into our shared human experience.
However, the reality of living with this profound neurological failure—especially when it comes to the unique horror of Cataplexy—is still profoundly misunderstood and unrecognized. Cataplexy is a sudden loss of muscle tone triggered by strong emotions, which is highly specific to NT1 and a core indicator of hypocretin deficiency [2][5].
Currently, many experts seem to hold a limited desire to acknowledge the full depth of the human experience, acting as though the reality of living with this disease is “completely and well understood”, suggesting “no need for evolving terminology and a broader breakdown into specifics and depth in regards to the symptoms”. While this criticism is not aimed at all medical professionals, as some remain very open-minded and continue to pursue better outcomes for persons living with Narcolepsy (PWN), a major disconnect persists around treatment.
So many seem to state that the “disease is treat-able”. Yet, they often simultaneously speak of the difficulties in finding the right treatments. The reality is that there are very many people who never find a successful treatment due to the wide, variable, and expansive nature of the disease spectrum. Even those who do find beneficial treatments tend to go through an ongoing juggle and battle with titration and/or switching medications. This failure to fully acknowledge the scope of treatment difficulties, much like perpetuating stereotypes such as calling excessive sleepiness “laziness” or cataplexy “weakness”, has profoundly negative and dauntingly impactful connotations and real-life effects for patients.
Yet, due to pervasive public stereotypes and a general lack of clarity and acknowledgment from the medical world, patients often endure a diagnostic delay of over a decade worldwide [1][2].
We are not lazy. We are fighting a systemic neurological failure every single day, often absorbing the negativity and dismissals of others who “choose to not see” the gravity of the situation.
For the sake of every person living with this condition – to ensure better clarity, comprehensive care, improved insights, comprehension, recognition, familiarity, and especially acknowledgment – the understanding of the NT1 human experience must evolve. We need to bridge the gap and disconnect that exist between the science and that lived human experience. Our symptoms, whether it’s cardiovascular dysfunction [1], depression, or a cataplexy episode that steals our physical control, deserve full acknowledgment, not minimization.
References
[1.] Klein G, Burghaus L, Vaillant M, Pieri V, Fink GR, Diederich N. Dysautonomia in Narcolepsy: Evidence by Questionnaire Assessment. J Clin Neurol. 2014 Oct 6;10(4):314–319. doi: 10.3988/jcn.2014.10.4.314.
[2.] Coelho FMS. Narcolepsy: an interface among neurology, immunology, sleep, and genetics. Arq Neuropsiquiatr. 2024 Apr 2;82(6):s00441779299. doi: 10.1055/s-0044-1779299.
[3.] Sheng D, Li P, Xiao Z, Li X, Liu J, Xiao B, Liu W, Zhou L. Identification of bidirectional causal links between gut microbiota and narcolepsy type 1 using Mendelian randomization. Sleep. 2024 Mar 1;47(3):zsae004. doi: 10.1093/sleep/zsae004.
[4.] Tafti M, Lammers GJ, Dauvilliers Y, et al. Narcolepsy-Associated HLA Class I Alleles Implicate Cell-Mediated Cytotoxicity. Sleep. 2016 Mar 1;39(3):581–587. doi: 10.5665/sleep.5532.
[5.] Chabas D, Taheri S, Renier C, Mignot E. The genetics of narcolepsy. Annu Rev Genomics Hum Genet. 2003;4:459–83.
[6.] Zhang R, Gao S, Wang S, Zhang J, Bai Y, He S, Zhao P, Zhang H. Gut Microbiota in Patients with Type 1 Narcolepsy. Nat Sci Sleep. 2021 Nov 6;13:2007–2018. doi: 10.2147/NSS.S330022.
Disclaimer: The information provided in this article is intended for informational and educational purposes only. Seek a qualified medical professional with expertise in Narcolepsy for diagnosis or treatment. I am not a medical professional.
Spanish version
Japanese/ソロモン・ブリッグスによる日本語訳
Created by: Solomon Briggs
(aka Narcoplexic)
October 23rd, 2025
The Unseen Dysfunction: Why Type 1 Narcolepsy is More Than ‘Just a Sleep Disorder’ © 2025 by Solomon Briggs is licensed under CC BY-NC 4.0
To view a copy of this license, visit https://creativecommons.org/licenses/by-nc/4.0/
“You may use this tool for non-commercial purposes, but must credit Solomon Briggs.”

